The ClpX Chaperone and Super-Resolution Imaging – a promising toolbox to explore Staphylococcus aureus cell division and the killing mechanism of β-lactam antibiotics
PhD student: Camilla Jensen
Thesis defended: 27 April 2021
BACKGROUND
You have probably taken penicillin to cure tonsillitis or other bacterial infections - but have you ever wondered how penicillin actually works?
Hundred years ago, most people died from pneumonia, tuberculosis, and other bacterial infections. Therefore, the discovery of penicillin and other antibiotics is one of the greatest breakthroughs in the history of medicine. Penicillin has saved millions (maybe billions!) of lives, but we still don’t know how this "wonder drug" kills bacteria.
In our research group, we work with the bacterium Staphylococcus aureus (S. aureus), which naturally inhabits the skin and nose of humans and animals. At the same time, this bacterium is one of the most common causes of both minor skin infections and life-threatening infections in, for example, heart valves and bones.
Normally, staphylococci are killed by penicillin, but by chance, we found a S. aureus mutant that surprisingly only was able to grow and multiply when we added penicillin to the growth media.
PURPOSE OF THE PROJECT
This is the first time it has been observed that penicillin can help a bacterial mutant multiply. We, therefore, hypothesized that this staphylococcal mutant could be the unexpected tool that could help solve the mystery of how penicillin works.
RESULTS
Initially, we wanted to investigate why the mutant cannot multiply in the absence of penicillin. Bacteria are tiny - they are typically smaller than 1/1000 of a millimeter. When we want to study bacteria, we therefore need powerful microscopes and within the last decade, advanced microscopes have been developed, which for the first time has allowed us to look all the way into the bacteria and visualize the smallest details. Using this type of microscopes, we found that the mutant has lost the ability to control the activity of enzymes that degrade the cell wall (“autolysins”). Normally, these enzymes are only activated when the bacteria are ready to divide, i.e. when it has built a cell wall across the cell. In the mutant, we on the other hand observed that the autolysins were activated prematurely causing the cell to burst because the immature cell wall was unable to withstand the internal pressure (Figure, top). The next step was to examine how penicillin helps the mutant to multiply. We found that when penicillin was added, the mutant was able to build a mature cell wall before activating the autolysins. Thus, penicillin helps the mutant multiply by delaying the autolysins and thereby preventing it from rupturing (Figure, bottom).
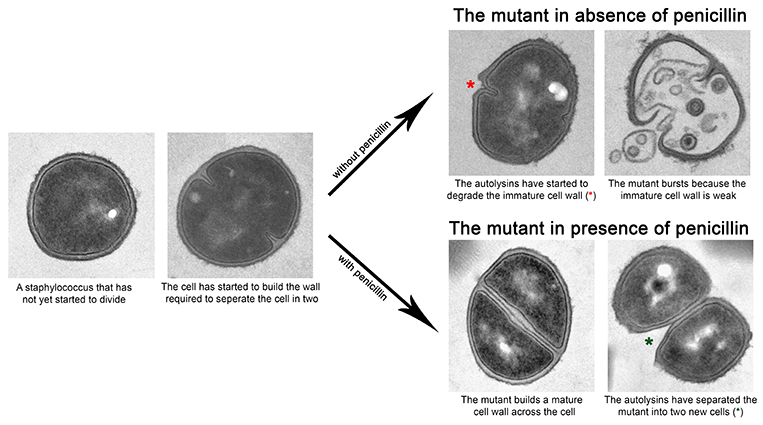
Our data, therefore, indicate that penicillin in normal circumstances inhibits the activation of the autolysins, which are important for the cell to divide and thereby prevents the bacterium from multiplying. With this study, we have come an important step closer to understanding how penicillin kills bacteria.
FUTURE PERSPECTIVES
Every year, between 60,000 and 100,000 Danes acquire a bacterial infection during hospitalization and therefore the increasing prevalence of antibiotic-resistant bacteria poses a particular threat in hospitals. In Denmark, MRSA (methicillin-resistant S. aureus) is one of the most common causes of life-threatening infections in debilitated patients. These infections are difficult to treat because the bacteria are resistant to exactly the type of antibiotic that the bacteria are best treated with - namely penicillin.
Nevertheless, it might become possible to treat MRSA infection with penicillin again – however, this requires identification of ‘helper-compounds’ that can restore the sensitivity of MRSA to penicillin. In this study, we additionally found that our staphylococcal-mutant can be used as a tool to find such ‘helper-compounds’. Using the mutant, we were able to specifically select compounds that rendered MRSA sensitive to penicillin in a library consisting of more than 50,000 random compounds. In the long run, these compounds have the potential to boost the effect of penicillin against MRSA and make treatment of resistant bacteria more efficient.
